Human trials of Vietnamese homegrown COVID-19 vaccine running smoothly
 |
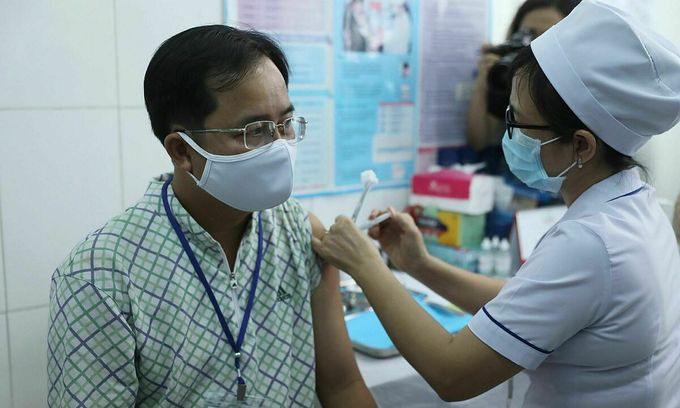
| A volunteer is about to be injected with Nanocovax at the Vietnam Military Medical University in the second phase. Photo: Vietnamnet |
A total of 560 people, aged from 12-75, have participated in the phase, jointly carried out by the Vietnam Military Medical University and HCMC Pasteur Institute in Hanoi and the southern province of Long An.
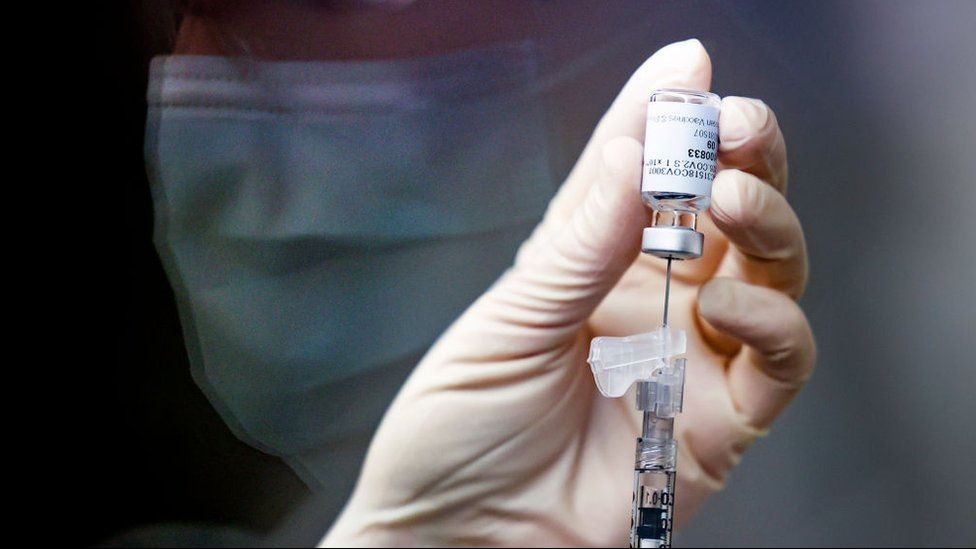
Of the volunteers, 367 have been injected since the phase began on February 26. The Vietnam Military Medical University alone has given shots to 120 volunteers, including 30 people above 60 years old.
The volunteers have injected 25 microgram, 50 microgram, and 75 microgram doses each. Following their shots, all volunteers would be monitored for 60 minutes. Afterward, they would be medically monitored by local authorities at home.
The second phase aims to allow researchers to see how exactly immunogenic Nanocovax is. Its preliminary results should be expected within May, said Do Quyet, Director of the Vietnam Military Medical University.
Afterwards, the Ethics Committee and the Ministry of Health would determine the most optimal doses for the third phase when the number of volunteers could reach between 10,000-15,000, he added.
 |
| A man in Long An Province is injected with Nanocovax, a Vietnamese Covid-19 vaccine, as part of the second phase of its human trials, February 26, 2021. Photo: VnExpress |
The first phase, which commenced last December with 60 volunteers, revealed Nanocovax to be safe, immunogenic and causing no severe side effects, according to Quyet.
Most of the volunteers of the first phase are in stable conditions after vaccination. Only few reported pain at the injection spot and fever that disappeared after one to two days.
Preliminary results also showed antibodies produced by Nanocovax had an effect on a coronavirus variant originating from the UK, which is up to 70 percent more transmissible than ordinary strains, he said.
 |
| A checkpoint at Pha Tai 2 village, Kim Dinh commune, Kim Thanh district of Hai Duong. Photo: VNA |
Developed by the Nanogen Pharmaceutical Biotechnology JSC, Nano Covax is the first Vietnam-made COVID-19 vaccine to enter human trials, with another two from other manufacturers to follow in February and March.
Vietnam is one of the 40 countries that have conducted human trials of a COVID-19 vaccine.
The country also has several other COVID-19 candidate vaccines being developed, by the Institute of Vaccines and Medical Biologicals (IVAC), the Company for Vaccine and Biological Production No.1 (VABIOTECH), and the Centre for Research and Production of Vaccines and Biologicals (POLYVAC)./.
| Six new cases of COVID-19 were reported in Vietnam in the past 12 hours to 6pm March 4, all in the northern province of Hai Duong, according to the National Steering Committee for COVID-19 Prevention and Control. The national tally increased to 2,488. The same day, 22 COVID-19 patients were given the all clear, bringing the total number of recoveries to 1,920. The death toll remains at 35. Among active patients, 65 have tested negative for SARS-CoV-2 once, 57 twice and 137 thrice. More than 51,570 people who had close contact with COVID-19 patients or arrived from pandemic-affected areas are under medical monitoring across the country. |
 | Oxford's new vaccine might be one of the strongest weapons against new Covid variants Amid the rising of the new Covid-19 variants that threat millions of lives, one of strongest weapons that might help curbing the spread of the ... |
 | Made-in-Vietnam COVID-19 vaccine to be priced under US $2.6 The indigenous Covivac vaccine is expected to be less than US $2.6 (VND 60,000) per dose, a reasonable price for all Vietnamese residents. |
 | Vietnamese Government issues resolution on COVID-19 vaccine purchase, use The Government on February 26 issued Resolution No 21/NQ-CP regarding the purchase and use of COVID-19 vaccines. |
Recommended
 Viet's Home
Viet's Home
Hung Yen - Japan: Strengthening Connectivity, Promoting Investment - Trade Cooperation, and Expanding Relations
 Viet's Home
Viet's Home
Gia Lai Allocates Over VND 2.1 Trillion for Safe Housing Projects
 Viet's Home
Viet's Home
Ho Chi Minh City And Italian Businesses Promote Strategic Cooperation
 Viet's Home
Viet's Home
Vietnam-Sweden Children's Film Week Opens in Hanoi
 Viet's Home
Viet's Home
Nguyen Anh Tuan Re-Elected President of the Vietnam General Confederation of Labour
 Viet's Home
Viet's Home
14th Vietnam Trade Union Congress to Elect 135 Members to New Executive Committee
 Viet's Home
Viet's Home
Vietnam and France Promote Parliamentary and Defense-Security Cooperation
 Viet's Home
Viet's Home